Otohiko TSUGE*, Taizo HATTA, Tomoko OHTO† and Kenichi SOMEKAWA†*
Department of Applied Chemistry, Faculty of Engineering, Sojo University; Ikeda, Kumamoto-shi 860-0082 Japan † Department of Applied Chemistry and Chemical Engineering, Faculty of Engineering, Kagoshima University; Korimoto, Kagoshima-shi 890-0065 Japan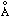 , and the other is longer, 2.4−3.4
, and the other is longer, 2.4−3.4 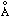 . The shorter TS distance may cause smaller activation energies. These phenomena seem to come from steric influence by two bulky substituents not to be coplanar to one another in 1a. Namely, steric hindrance of two substituents in the 1,3-dipolar seems to cause asymmetric TS and to derive two-step reactions of lower activation energy.
. The shorter TS distance may cause smaller activation energies. These phenomena seem to come from steric influence by two bulky substituents not to be coplanar to one another in 1a. Namely, steric hindrance of two substituents in the 1,3-dipolar seems to cause asymmetric TS and to derive two-step reactions of lower activation energy.